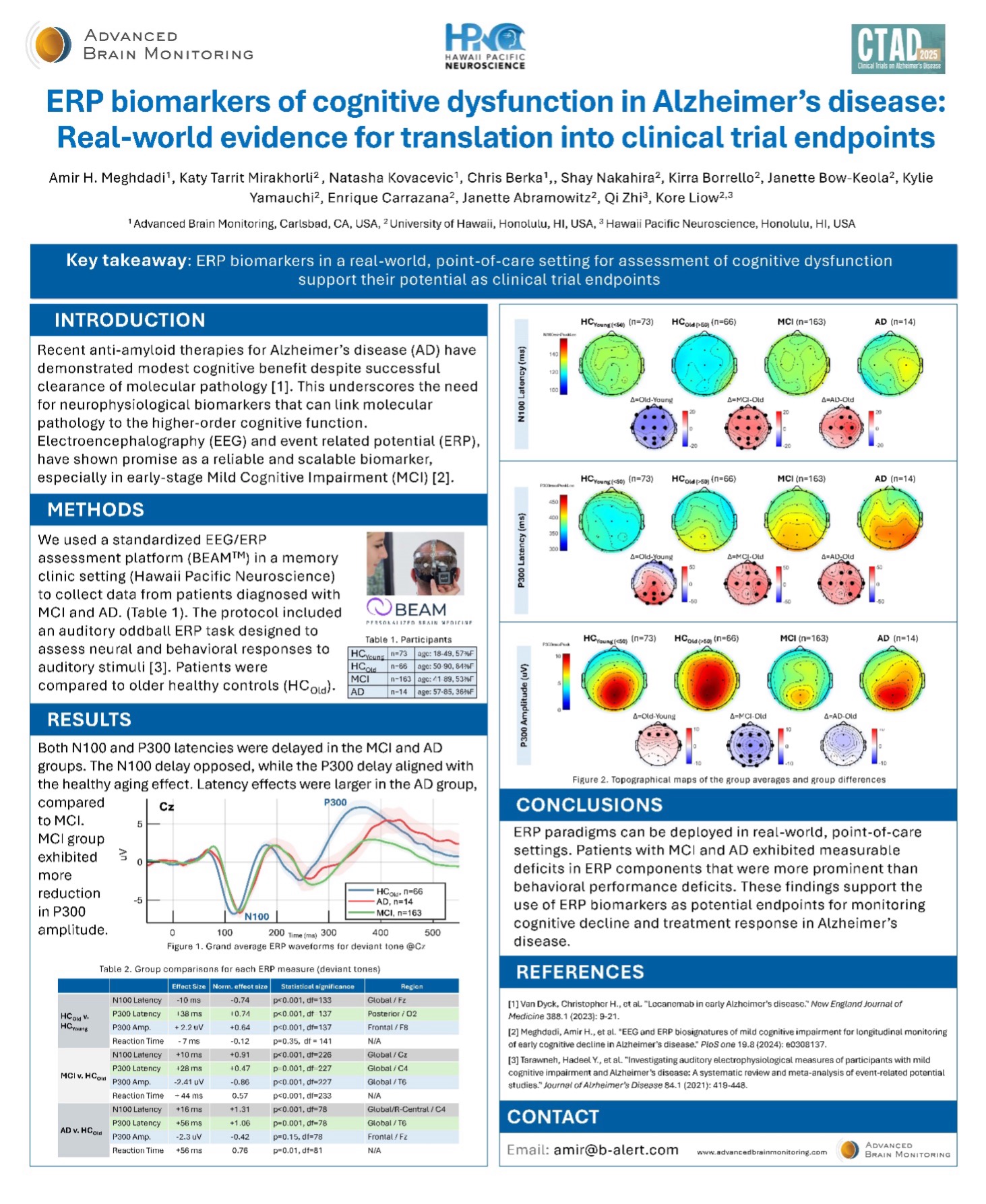
ERP Biomarkers of Cognitive Dysfunction in Alzheimer’s Disease: Real-World Evidence Supporting Translational Clinical Trial Endpoints
Advances in neuroscience and digital biomarkers are transforming how clinicians and researchers detect, monitor, and study Alzheimer’s disease (AD). At the 2025 Clinical Trials on Alzheimer’s Disease (CTAD) Conference in San Diego, researchers presented compelling evidence demonstrating how event-related potentials (ERP) derived from electroencephalography (EEG) can serve as reliable, real-world biomarkers of cognitive dysfunction and meaningful translational endpoints for clinical trials.
This work highlights the growing role of ERP biomarkers in bridging laboratory neuroscience with real-world clinical application.
Understanding Cognitive Dysfunction in Alzheimer’s Disease
Alzheimer’s disease is characterized by progressive cognitive decline affecting memory, attention, executive function, and information processing. Traditional diagnostic and monitoring tools—such as neuropsychological testing, imaging, and fluid biomarkers—are valuable but can be costly, invasive, or limited in capturing real-time brain function.
Electrophysiological measures, particularly EEG and ERP, offer a complementary approach by directly measuring neural activity with high temporal resolution. ERP responses reflect how the brain processes cognitive stimuli, making them especially useful for identifying subtle changes in cognitive function.
What Are Event-Related Potentials (ERP)?
Event-related potentials are time-locked EEG signals generated in response to specific sensory or cognitive events, such as memory tasks or attention cues. ERP components are characterized by their latency (timing) and amplitude (signal strength), both of which can be altered in neurodegenerative disease.
In individuals with:
- Mild Cognitive Impairment (MCI)
- Alzheimer’s disease (AD)
ERP patterns often show delayed responses and reduced signal amplitudes, reflecting impaired cognitive processing.
A key strength of the research presented at CTAD 2025 is its focus on real-world ERP data, collected in practical clinical environments rather than highly controlled laboratory settings. This approach demonstrates that ERP biomarkers remain reliable and informative even outside traditional research conditions.
The findings show:
- Consistent ERP differences between cognitively normal individuals, MCI patients, and Alzheimer’s patients
- Measurable changes in brain responses linked to cognitive dysfunction
- Strong reproducibility across real-world clinical settings
This real-world validation is critical for broader clinical adoption.
ERP as a Translational Clinical Trial Endpoint
One of the major challenges in Alzheimer’s research is identifying sensitive, scalable, and clinically meaningful endpoints for therapeutic trials. ERP biomarkers address this challenge by offering:
- Non-invasive measurement of brain function
- Objective physiological data, independent of patient self-report
- High sensitivity to cognitive change
- Repeatability, making them ideal for longitudinal studies
The evidence presented supports ERP measures as translational endpoints, capable of linking underlying neural changes to clinical outcomes in Alzheimer’s disease trials.
The integration of ERP biomarkers into clinical trial design may significantly improve how researchers:
- Detect early cognitive changes
- Monitor disease progression
- Evaluate treatment response
- Reduce reliance on invasive or costly biomarkers
By capturing real-time neural processing, ERP measures can enhance the precision and efficiency of clinical trials while supporting earlier intervention strategies.
This research underscores the growing importance of digital and neurophysiological biomarkers in modern neurology. EEG-based ERP tools offer a scalable solution that can be deployed in point-of-care and research settings, supporting personalized medicine approaches in Alzheimer’s disease.
As Alzheimer’s research continues to evolve, ERP biomarkers represent a promising pathway toward more accessible, objective, and clinically meaningful measures of cognitive dysfunction.
Conclusion
The real-world ERP evidence presented at CTAD 2025 strengthens the case for event-related potentials as powerful biomarkers of cognitive dysfunction in Alzheimer’s disease. By demonstrating reliability outside the laboratory and relevance to clinical outcomes, this work supports the adoption of ERP measures as translational endpoints in Alzheimer’s clinical trials.
These findings mark an important step toward improving early detection, monitoring disease progression, and accelerating therapeutic development for individuals affected by Alzheimer’s disease.