The Hawaii ALS and Neuromuscular Center has been selected to participate in the ADAPT-EARLY clinical trial, a Phase 4 research study evaluating efgartigimod PH20 SC in adults with new-onset generalized myasthenia gravis (gMG). This important study focuses on early treatment intervention for individuals diagnosed with gMG within the past year .
What Is Generalized Myasthenia Gravis?
Generalized myasthenia gravis (gMG) is a rare, chronic autoimmune neuromuscular disease that affects voluntary muscle strength. The condition is caused by immunoglobulin G (IgG) autoantibodies that disrupt communication at the neuromuscular junction, leading to impaired muscle contraction and weakness .
Common symptoms of gMG include:
- Muscle weakness that worsens with activity
- Difficulty walking or lifting objects
- Slurred speech and swallowing problems
- Drooping eyelids or double vision
- Breathing difficulties in severe cases
Early diagnosis and treatment are critical for improving long-term outcomes and quality of life.
About the ADAPT-EARLY Study
The ADAPT-EARLY study is a Phase 4, open-label, prospective, single-group, multicenter clinical trial designed to evaluate the effectiveness of efgartigimod PH20 SC in adults with newly diagnosed generalized myasthenia gravis .
Study Details:
- Participants: Adults with gMG symptoms for less than one year
- Treatment: Subcutaneous efgartigimod PH20 SC
- Study Duration: Approximately 58 weeks total
- Treatment Period: 51 weeks
- Objective: To assess clinical response to efgartigimod in early-stage gMG patients
This study aims to better understand how early use of targeted therapies can improve disease management.
How Efgartigimod Works in gMG
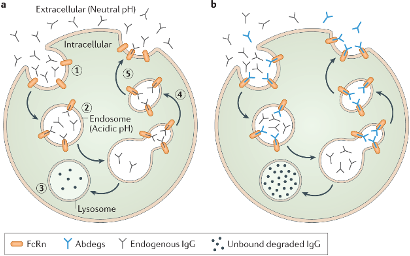
Efgartigimod is a first-in-class therapy that targets the neonatal Fc receptor (FcRn). By blocking FcRn, the medication prevents the recycling of IgG antibodies, leading to reduced levels of disease-causing autoantibodies in the body .
Lowering IgG levels may help:
- Improve neuromuscular transmission
- Reduce muscle weakness
- Improve daily functioning and quality of life
Expert Leadership in Neuromuscular Research
The study is led by Natalia Gonzalez, MD, Director of the Hawaii ALS and Neuromuscular Center and Principal Investigator of the Neuromuscular Research Unit. Dr. Gonzalez is dual fellowship-trained in Neuromuscular Medicine and Neuroimmunology and serves as a Clinical Assistant Professor of Neurology at the University of Hawaiʻi John A. Burns School of Medicine .
Learn More About the gMG Clinical Trial in Hawaii
Patients or caregivers interested in learning more about this generalized myasthenia gravis clinical trial in Hawaii may:
- Visit official NIH clinical trial resources
- Call the Dedicated Neuromuscular Research Hotline: (808) 564-6141
